Molecular mass (molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u). (1 u is equal to 1/12 the mass of one atom of carbon-12) Molar mass (molar weight) is the mass of one mole of a substance and is expressed in g/mol. Weights of atoms and isotopes are from NIST article.
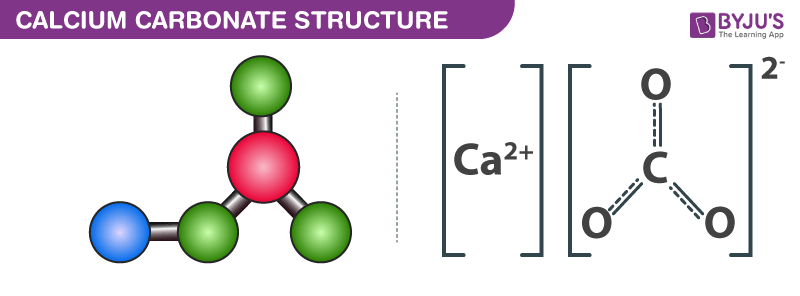
Solving for the atomic mass of calcium carbonate (CaCo3)
- Answers Chemistry GCSE Article Calculate the relative molecular mass of CaCO3. (relative atomic mass (Ar): C= 12, Ca= 40, O= 16).
- 1000g CaCO3 x 1mol CaCO3/108.09g CaCO3=9.252 mol CaCO3. 9.252mol CaCO3 x 1mol CaO/1mol CaCO3= 9.252 mol CaO. 9.252 mol CaO x 64.08g CaO/1mol CaO= 592.9g CaO. 592.9g= 0.5929kg. The answer in kg is 0.5929 kg CaO. 108.09 is the molar mass of CaCO3 and 64.08 is the molar mass of CaO.
- Atomic mass of Ca is 40, of C is 12 and of O is 16. Therefore molecular mass of CaCO3 is 40+12+3(16) which is 100. Percentage composition of Ca by mass is 40 x 100/100 which is 40%. Percentage composition of C by mass is 12 x 100/100 which is 12%. Percentage composition of O by mass is 48 x 100/100 which is 48%.
Need to know the atomic mass of a calcium carbonate molecule? Our molar mass calculator uses the periodic table and the chemical formula to solve for the molar mass of a chemical compound based on the compound's empirical formula. The calculator takes the elemental composition of the compound and weighs the elements to get an empirical formula mass. Note that the calculator assumes a pure substance - if you're aware of dilution or impurities, make appropriate adjustments for the molarity of a given substance.
This project started with as a molar mass calculator for chemical reactions. You can use our calculator to solve for the theoretical yield of an experiment. We also have a percent yield calculator which can help you apply this to actual experiments. Use the mole ratio and empirical formula to understand the limits of the reactants.
Other terms: atomic mass of calcium carbonate, molar mass of calcium carbonate, molecular mass, Happy birthday ankit song download.
How Does The Molar Mass Calculator Work?
We take the formula you provide (NaCl - common table salt - in our default example) and unpack it into the component elements. Then we compare each atom against a table of the standard atomic weights for that element. We present the results in a table at the bottom of the molar mass calculator - it will show the count of atoms, the atomic weight of each element, and the molecular weight for the molecule. It solves for total mass of a molecular formula (average molecular weight). Kepler 47a.
From there we break the formula for calcium carbonate into parts - a Calcium atom, a Cobalt atom, etc.
We don't have brackets implemented (yet), so you will need to unpack any bracketed expressions. They don't affect the weight anyhow. Simply take each element and multiple it by the number of times the bracketed structure occurs. For example: (C6H5)3PCCO => C18H15PCCO
Finding Molar Mass for Other Chemical Compounds
Our molar mass calculator has this for a variety of other compounds: sodium chloride, carbon dioxide, sulfuric acid, glucose..
Bookmarking, Save, and Share Results
The tool is designed so you can flip between different parts of a problem set. We recommend you bookmark it so you can refer back to it. You can also share results with a study partner or tutor by hitting calculate and copying the URL for this page. When your study partner opens up the URL, they will see your calculations. It's easy share & save results via email. (Be sure to hit calculate first, however)
You also have the option of saving links to the calculations in your research notes files, so you can quickly re-open or check them later. Again - hit calculate first so the URL is updated with your most recent changes. Then copy and save the url.
FAQ - Molar Mass Calculator
What is Molar Mass in Chemistry?
What Is The Atomic Mass Of Caco3
Molar mass is an important concept in adapting chemical formulas to real world conditions. We may be able to balance a chemical equation and determine that one molecule of hydrogen combines with two of oxygen to make water (or the compound of your choice). But how would you set up the materials in the laboratory? Or if you were, for example, buying oxygen for a process, how would you determine how much to use to make a given quantity of water? Molar mass allows us to convert a chemical reaction into specific amounts of reagents required for the process. By converting the atomic interaction into grams, we can measure and use an appropriate amount of the necessary reagents. Formula mass helps us solve for this.
What Is Relative Atomic Mass / Relative Molecular Mass / Average Molecular Weight?
Co pay relief fund. A copay, short for copayment, is a fixed amount a healthcare beneficiary pays for covered medical services. The remaining balance is covered by the person's insurance company.
The relative atomic mass of a compound is the ratio of the average mass of the elements in a chemical compound to the atomic mass constant, which is defined as 1/12 the mass of a carbon 12 atom. For a single sample, the relative atomic mass of the sample is the weighted arithmetic mean of the masses of the individual atoms present in the sample (also known as the average atomic mass). This will vary by isotope of the element (carbon-12 vs. carbon-13, for example, since the two isotopes have a different atomic mass due to additional neutrons). In the real world, this can vary based on where the sample was collected - due to variances in the specific isotopes of the elements present (driven by differences in radioactive decay and how the material was aggregated to begin with).
How To Find Molecular Mass Of Caco3
How to find Molar Mass
Molar Mass Of Caco3
Take a standard chemistry formula for a molecule, split it up into the component atoms, and look up the molar weight of each atom. Add the weight of the atoms in the molecule and you have the molar mass for the molecule.
